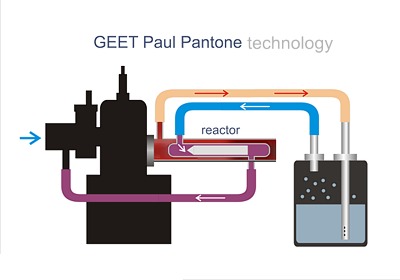
GEET is a popular technology for using water as a fuel. How is this possible to consider an example. Take 10 liters of gasoline, pour into the tank. We start the engine and went for a drive. We're driving until the gasoline runs out. Question: how much did the engine use? Think 10 liters? Not guessed right answer is 8 liters. And where is the rest of the fuel? The rest of the fuel burned the catalytic converter (catalyst). The catalyst has the following purpose: to burn everything that has not burnt in the cylinders. And only 70-80 percent of hydrocarbons burn in cylinders. That's where the reserve for saving fuel is hidden. If you do not resort to techniques on the verge of insanity, such as a special prayer or an image in a cigarette lighter, the real way to save fuel is to return the unburned gasoline back to the cylinders rather than the useless afterburning of such a valuable product in the catalyst. In exhaust except for gasoline there is soot, and it can also be fuel. In addition, there is a lot of water in the exhaust. All this can be put into useful work. It is known that water (H2O) can be used as fuel if it is decomposed into hydrogen and oxygen. This is done either by electrolysis or by heating to a high temperature. In the latter case, the HHO mixture is called Brown gas. Such a mixture can not exist for long. It is an excellent "rattler", not worse than dynamite. At high temperatures, H2O molecules decay, but when the mixture is cooled, the atoms instantly combine into molecules. If in the electrolysis the components are obtained separately, a high-temperature decomposition, and then subsequent cooling, causes an explosion. These components obtained in the high-temperature decomposition can only be stored in a mixture with nitrogen (N) or carbon dioxide (CO2), which do not support combustion. Ways of using water as a fuel are well known. For example, for already four decades in Vinkler generators, an excellent industrial fuel is produced which is a mixture of CO carbon monoxide with hydrogen, obtained by reacting heated to 600 degrees of coal with steam according to the reaction: С + Н2О = СО + Н2 (1) Hydrogen is easily separated from the monoxide. One of the methods is as follows: inside the steel tube, which is heated in the furnace, another thin metallic (copper, silvered) is placed, through which a constant jet of cold liquid flows. Carbon monoxide, coming in contact with a heated surface, gives oxygen to the iron. The released carbon forms coal, and its particles sit in the form of soot on a cold tube. In turn, carbon dioxide can be converted to monoxide by passing it through coal. This is explained by the fact that carbon atoms, when coal is heated with carbon monoxide CO2, acquire an affinity: CO2 + C = 2CO (2) Oxygen seems to be redistributed among all the carbon atoms available in the composition of CO2 and in the composition of coal. Mole CO2 and mole C give two moles of CO. For the disintegration of water, there are many patents. For example, the patent (19) SU (11) 64434 (13) A1. The patent was declared as early as March 22, 1943. The essence of the patent: to protect the fuel tanks from explosions and fire, as well as to increase the altitude of the petrol system of aircrafts, filling gasoline tanks with neutral gaseous components is used. The exhaust gases of the engines contain a water slurry and cool, form a condensate, when freezing, the normal operation of the petrol system is disrupted. It is proposed to apply the method of decomposition of suspended matter for the dehumidification without the release of condensate. The exhaust gases of the motors under normal operating conditions have a temperature of about 600-700 degrees. Drops and molecules of H2O, in contact with coal, decompose almost completely, according to reaction 1. For complete dehydration it is proposed to use iron, which is first oxidized and then reduced by monoxide. Of course, we do not propose to carry coal in the trunk for the sake of fuel economy. The foregoing only demonstrates that the thermal decomposition of non-combustible water into combustible components is not at all fiction, as many think. In addition, in reactions it is easy to get not only hydrogen, but also carbon monoxide, which, in terms of energy value, is virtually inferior to hydrogen, but does not explode. Recently, GEE technology has been very popular. Its feature is an additional application of the electric field. Moreover, the field arises as a result of the friction of atoms over metal, which excludes the use of an external high-voltage source. The inventor of the technology is Paul Pantone, although the use of an electric field for the decomposition of H2O has not been news for a long time. There are many patents on this subject. Panton's merit is more in the popularization of technology, bringing it to the minds of the masses, for which the inventor gained fame and a lot of trouble. The Pantone Reactor is a steel tube with a pointed core inside. The vapor, passing through the gap, decomposes into components. At the same time, the tube receives hydrocarbons, as well as carbon dioxide and nitrogen, which eliminates the recombination of hydrogen. However, some oxygen reacts either with hydrocarbons, or with carbon monoxide, or with hydrogen. The electric field improves the reactions, and makes them possible with cooler components. Decomposition does not require the supply of energy. The device presented is not super-unitary, but the heat emitted into the exhaust pipe is essentially a dummy, because it is a by-product of the engine. Accordingly, the device utilizes thermal energy. The constructions of such devices are great. Each experimenter, making an assembly with his own hands, considers it his duty to make changes. Often these changes lead to inoperability. For example, take and fix the rod on a metal extension, after which the static charge goes away and the electric field does not arise. Or they extend the rod beyond measure, and the channel too strongly resists. Or increase the gap, weakening the field strength. Despite the apparent simplicity, the design requires scrupulous precision in manufacturing. Otherwise, the device works as a simple "water heater". When operating a car in a dry climate, water injection is sufficient to save 10-15%. But if your street is always raining, do not expect a special effect. The classical Pantone scheme is given below. Exhaust gases, through a branch in the exhaust pipe, enter the tank with water - a burbulant. The suspended aqueous slurry enters the reactor through a hose. The output of the device is connected to the intake manifold of the internal combustion engine.
Separate problem - burbulyator. Its task is to bring the liquid in the flow tank to a vapor state. Suspension in an acceptable amount, appears only after warming the tank, and this process is very inert. As a result, the system is single-mode, no effective application of it on the car and there can be no talk. And yet an acceptable solution for the car exists. The solution is to abandon the tank with the burbulant, and take everything that is needed directly from the exhaust line. For every liter of burned fuel, a car produces a third of a liter of water. For the normal operation of the construction, only half is needed. It is possible to separate water in a vortex centrifugal chamber using the fact that the H2O molecules are slightly lighter than nitrogen molecules, and the drops are much heavier than nitrogen. Thus, the technology acquires a new quality. It is not necessary to think that it is too easy to separate the useful from the useless in the vortex chamber. Thus, in the classical Ranka tube, the separation of molecular components does not occur. However, if we combine the twist with the throttling, we come to the fact that water and hydrocarbons will be extracted with an efficiency of more than 70%. Even if you exclude the reactor from the system - just supply steam and unburned hydrocarbons in the cylinders with a pit in the intake manifold, it is easy to achieve savings of 10-20 per cent. An analogy with water injection systems is remotely traced, with the difference that a water tank will not be needed. In addition, the engine will add to the torque and will be less heated, which is especially important with air cooling. In addition, the noise of the car will decrease, and the exhaust will become cleaner.
|
|
| Original DIY or buy |